Introduction
Hydroxytyrosol is a naturally occurring polyphenol primarily found in olives and their products, such as olive oil and olive leaves. It is widely studied for its outstanding antioxidant properties and has numerous applications in the food and cosmetic industries. As a powerful antioxidant, hydroxytyrosol helps reduce oxidation in foods, extending shelf life and preserving flavor. Due to its health benefits, hydroxytyrosol is often added to health foods to support cardiovascular health, boost immunity, and reduce inflammation.
- Common name (trade name): Hydroxytyrosol;
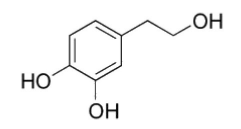
- Chemical name: 3,4-Dihydroxyphenylethanol;
- Molecular formula: C8H10O3;
- Molecular mass: 154.165 g/mol (according to the 2022 International Standard Relative Atomic Mass); and
- Structural formula:
This article analyzes the regulatory status of hydroxytyrosol in China, Europe, the United States, Australia, and New Zealand from a food industry perspective.
Approval status of hydroxytyrosol in China, Europe, the US, Australia, and New Zealand
Country/region | Approval status | Substance | Date of approval | Applicant |
China | Approved as a new food additive (antioxidant) | Hydroxytyrosol | July 25, 2024 | / |
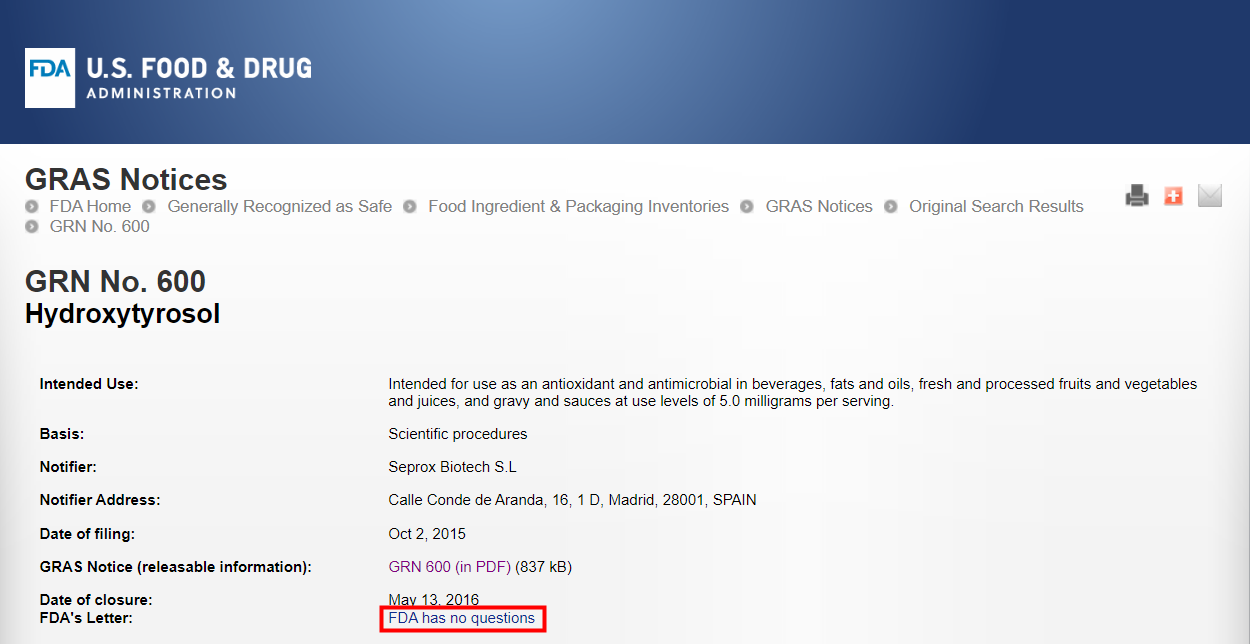
US | FDA GRAS (GRN No.600) | Hydroxytyrosol (chemical synthesis) | October 2, 2015 | Seprox Biotech S.L |
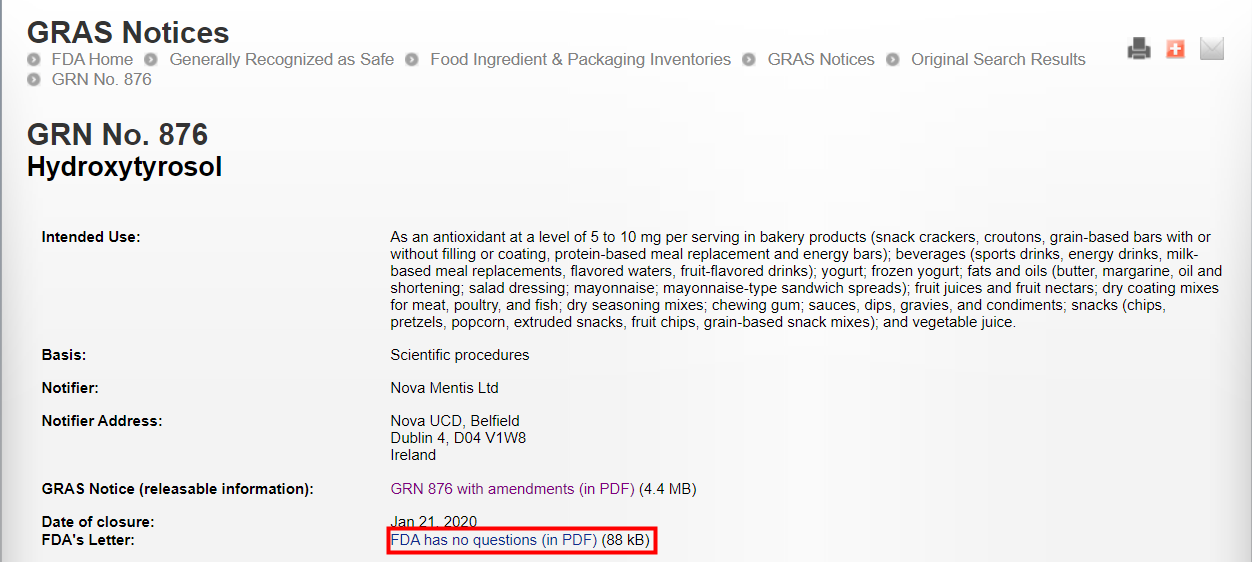
FDA GRAS (GRN No.876) | Hydroxytyrosol (biosynthesis) | January 21, 2020 | Nova Mentis Ltd | |
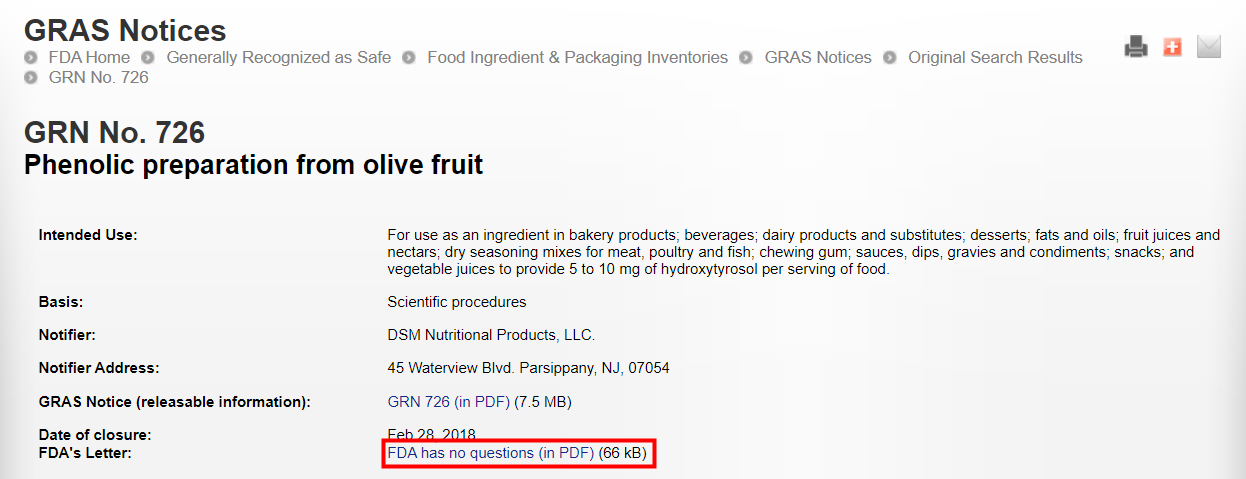
FDA GRAS (GRN No.726) | Phenolic preparation from olive fruit | February 28, 2018 | DSM Nutritional Products, LLC. | |
FDA GRAS (GRN No.978) | Hydrolyzed aqueous olive pulp extract | December 10, 2021 | Oliphenol LLC | |
FDA GRAS (GRN No.1138) | Hydroxytyrosol | Pending | Hangzhou Viablife Biotech | |
EU | Novel Food | Hydroxytyrosol (chemical synthesis) | December 14, 2017 | Seprox Biotech |
Novel Food | Olive fruit dry extract standardized in hydroxytyrosol (10% or 20%) | Ongoing Risk Assessment | NATAC BIOTECH S.L. | |
Australia and New Zealand | Food/Food Ingredients Record of Views | Olive fruit extract | Officially published in 2014 | / |
China
Hydroxytyrosol has been approved as a new food additive in China. It is used as an antioxidant in vegetable oils (food category 02.01.01) to delay oxidation, with a maximum level of 0.05 g/kg. This approval applies to hydroxytyrosol produced from tyrosine through a process involving fermentation with Corynebacterium glutamicum, followed by catalytic conversion, separation, and purification.
The approval process spanned just over six months from acceptance to public consultation, and only about nine months to receive official approval.
September 25, 2023 |
|
October 18, 2023 |
|
March 13, 2024 |
|
July 25, 2024 |
|
US
In the US, hydroxytyrosol has been approved as a Generally Recognized as Safe (GRAS) substance. Notably, another GRAS substance is hydrolyzed aqueous olive pulp extract, a phenolic preparation. Hydroxytyrosol is primarily intended for use as an antioxidant and antimicrobial in beverages, fats and oils, fresh and processed fruits and vegetables and juices, and gravy and sauces at use levels of 5.0 milligrams per serving.
EU
In accordance with Commission Implementing Decision (EU) 2017/2373, hydroxytyrosol derived from chemical synthesis has been authorized as a Novel Food. The authorized food categories include fish and vegetable oils (excluding olive oil and olive pomace oil), as well as spreadable fats, with maximum levels set at 0.215 g/kg and 0.175 g/kg, respectively. Additionally, the European Food Safety Authority (EFSA) has issued a scientific opinion on the safety of hydroxytyrosol as a Novel Food. The opinion states that the expected daily intake of hydroxytyrosol from these uses is comparable to or even lower than the exposure from consuming olive oil and olives. Therefore, the panel concluded that hydroxytyrosol is safe under the specified uses and use levels.

Australia and New Zealand
According to the “Food/Food Ingredients Record of Views” by Food Standards Australia New Zealand (FSANZ), the safety assessment for olive fruit extract notes that there are no safety concerns identified at levels of use (up to 20 mg/day of hydroxytyrosol). Therefore, it can be concluded that a daily intake of 20 mg of hydroxytyrosol is considered safe in Australia and New Zealand.

If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.
Further Information
CIRS Group Assists Client in Securing Approval for New Antioxidant Hydroxytyrosol
About the CIRS Global Food Ingredients Application Services
The CIRS food team offers the following services:
- Genetically Modified Microorganism New Food Raw Material Application
- Genetically Modified Microorganism Food Additive Registration in China
- US FDA GRAS Application
- US New Dietary Ingredient (NDI) Application
- EU Novel Food Application
- Novel Food Application in Australia and New Zealand
- EU Enzyme Preparation Application

